Chemical Equation For Co2 And Lime Water
The lime water test is used to detect the carbon dioxide gas. 22032019 Chemical Equation For The Reaction Of Co2 And Lime Water Ca Oh 2 Tessshlo Ca Oh 2 Co2 Caco3 H2o Balanced Equation Calcium Hydroxide Carbon Dioxide Carbonate Water You What Is The Word Equation Of Limewater Reacting With Carbon Dioxide Quora.
When Is The Excess Of Carbon Dioxide Passed Through Lime Water Quora
B CaO s H 2 O Ca OH 2 heat.

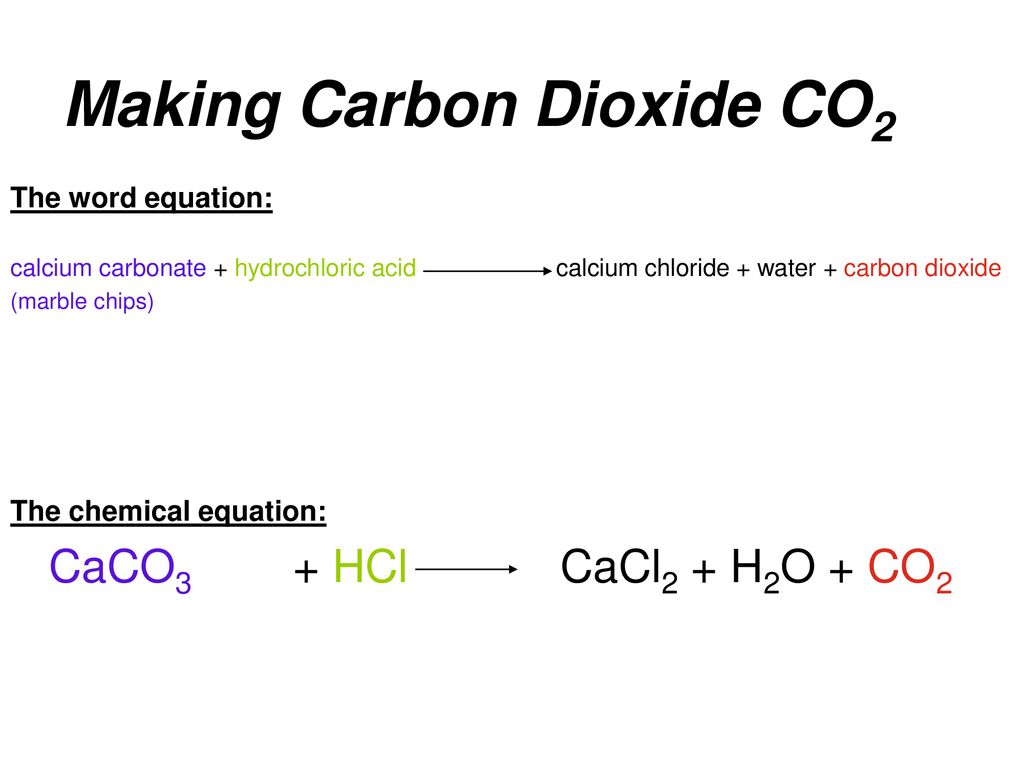
Chemical equation for co2 and lime water. If CO2 gas is passed in excess amounts then the above reaction occurs further. CaCO 3 s limestone CaOs lime CO 2 g carbon dioxide. Answer- B formation of water soluble calcium bicarbonate.
E CO2 Emissions of CO 2 in. On passage of carbon dioxide the lime water turns milky. CaCO 3 s H 2 O l CO 2 g CaHCO 3 2 aq.
The following reaction takes place. 22102020 The solution of lime water turns milky due to the formation of calcium carbonate which is a white precipitate insoluble in water. This also suggest that for each mgL of.
Approach 1 is based on the following equation. H2CO 3 Ca OH 2 CaCO 3 2H2O 1 Eq1 is the neutralization reaction between CO 2 carbonic acid and lime. In its equation form that makes.
19072018 Chemical Equation For The Reaction Of Co2 With Lime Water Tessshlo Balanced Equation For The Reaction Of Carbon Dioxide And Lime Water Tessshlo Ca Oh 2 Co2 Caco3 H2o Balanced Equation Calcium Hydroxide Carbon Dioxide Carbonate Water You. Continue adding the carbon dioxide until a colour change is observed. E Q SR CaO H H O CF CO2 i i i i i 1 2 Where.
Written as an Equation. 05082015 The reaction between limewater which is a solution of calcium hydroxide CaOH_2 and carbon dioxide will result in the formation of an insoluble solid called calcium carbonate CaCO_3. Lime water is the name for Calcium hydroxide.
Lime calcium oxide is a white solid with strongly basic properties. CaOH_2aq CO_text2aq -. The formula for lime water is CaOH 2 and the chemical name for lime water is calcium hydroxide.
24072017 Asked by Topperlearning User 24th Jul 2017 0137. Resultant carbon dioxide passed through limewater in the right tube producing a milky solution due to precipitation of the insoluble suspension of calcium carbonate. 14072018 Lime Water Formula.
Place about 125 cm 3 of water in a 250 cm 3 conical flask. When water is added to lime calcium hydroxide CaOH 2 is formed according to the following reaction. Is passed through lime water it turns milky due to the formation of calcium carbonate which is sinsoluble in water.
01072018 a Write chemical equations. Add one or two drops of phenol red to the water. CaO H 2 O CaOH 2.
CaOH 2 aq CO 2 g CaCO 3 s H 2 O l If excess CO 2 is added. 15092015 What happens when you mix carbon dioxide and lime water. A When quick lime is added to water it reacts to form calcium hydroxide slaked lime.
CO2 Ca OH2 ----. CaCO3 H2O CO2 Ca HCO32. Talk or blow gently into the flask ie add the carbon dioxide.
CaCO_3s darr H_2O_l The solution will turn milky due to the fact that calcium carbonate is a white precipitate. Its water that contains a higher-than-normal amount of calcium carbonate or calcium sulfate. B List two natural forms of calcium carbonate.
Ii When excess of carbon dioxide gas is passed through lime water. Lime is added to raise the pH while sodium carbonate is added to provide a source of carbonate ion. A considerable amount of heat energy is released during this reaction.
Finally the emission estimates are corrected for the production of any hydrated lime and any uncalcined Lime Kiln Dust LKD that is not recycled to the kiln. I When carbon dioxide gas is passed through lime water. This equation does not result any net change in water hardness.
This reaction is strongly exothermic and takes place vigorously with the formation of clouds of steam. Add two drops of sodium hydroxide solution to produce a red solution. Lime itself is.
Lime reacts readily with water to produce slaked lime which is the chemical compound calcium hydroxide. CaOH2 CO2 -. Is passed through lime water.
Specific type of lime. The second variety is manmade. Milk of lime Its a solution made from lime acted upon or slaked by water.
CaCO3 is insoluble in water and hence precipitates out in the solution thus making the solution look milky. 18092017 The first is natural.
Why Does The Milkiness Disappear From Limewater When Excess Co2 Passes Through It Quora

Carbon Dioxide Test Stock Image A500 0279 Science Photo Library
How Does Limewater React To Carbon Dioxide What Causes This Reaction Quora

How Does Limewater React To Carbon Dioxide What Causes This Reaction Quora

Pin On Middle Science For Seed

Limewater Test To Check For Carbon Dioxide In Your Breath Videos Examples Activities

Write The Chemical Equation Of The Reaction Taking Places When Co2 Gas Is Passed In Excess Through Brainly In

Test For Carbon Dioxide Aqa Gcse Chemistry Revision Notes
Post a Comment for "Chemical Equation For Co2 And Lime Water"