Chemical Formula For An Ionic Compound Represents
12052021 The chemical formula for an ionic compound represents one. In the formula of an ionic compound the sum of the ionic charges in the formula is.
02012021 A stable ionic compound is electrically neutral where electrons are shared between cations and anions to complete outer electron shells or octets.

Chemical formula for an ionic compound represents. The chemical formula for an ionic compound represents one formula unitthe simplest ratio of the compounds positive ions cations and its negative ions anions. Since it represents only one unit of the vast NaCl array or lattice. 15052020 Composition of ionic compounds.
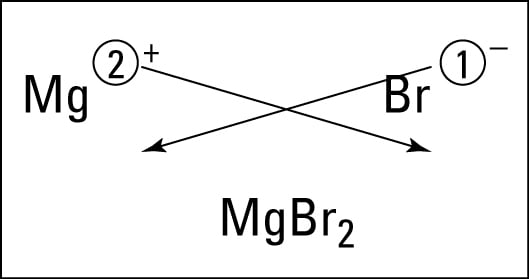
More accurately Al2S3 is the chemical formula for the ionic compound that is formed when the aluminum ion Al3 the cation formed when aluminum ionizes and the sulfide ion S2 the anion formed upon the ionization of sulfur bond with one another. This chemical formula says that there are one magnesium ion and two chloride ions in this formula. You know you have the correct formula for an ionic compound when the positive and negative charges on the ions are the same or cancel each other out.
When an ionic compound is formed from magnesium and oxygen the magnesium ion has a 2 charge and the oxygen atom has a 2 charge. 04062020 Polyatomic ions are enclosed in parenthesis followed by a subscript if more than one of the same ion exist in a chemical formula. For an ionic compound.
The electron configuration of nitrogen is 1s22s22p3. Sodium has a positive 1 charge and chloride has a negative 1 charge. Number of ions present in a sample.
How many more electrons does nitrogen need to have an electron configuration similar to neon. The chemical formula of an ionic compound shows the. 13082020 We could write the chemical formula for this ionic compound as ceMgClCl but the convention is to use a numerical subscript when there is more than one ion of a given typeceMgCl2.
The molecular formula gives the number of atoms of each element contained in a single molecule of the compound Chemical formulas are important because they indicate the relative. Independent neutral unit that can be isolated and examined. The chemical formula for aluminum sulfate an ionic compound consisting of aluminum cations and polyatomic sul- fate anions is written as shown on page 204.
The complete ionic equation is K aq Br aq Ag aq C 2 H 3 O 2 aq K aq C 2 H 3 O 2 aq AgBrs. The chemical formula for aluminum sulfate an ionic compound consisting of aluminum cations and polyatomic sulfate anions is written as shown on the next page. 18062020 For any ionic compound that is aqueous we will write the compound as separated ions.
What is the empirical formula for a compound that contains 1262 Li 2917 S and 5821 O. Chemical formulas represents one formula unit Formula unit. The formula for sodium chloride is NaCl - it shows that for every Na ion there is one Cl- ion.
Total number of ions in the crystal lattice. Tap again to see term. An ionic compound is mainly composed of.
Simplest ratio of ions that results in an electrically neutral compound d. The formula of an ionic compound shows the relative numbers of the ions it contains. The chemical formula for an ionic compound represents the simplest ratio of ions that results in an electrically neutral compound.
The chemical formula for an ionic compound represents a _____ _____ not a _____. This formula is called the formula unit. Therefore the proper formula for this ionic compound is MgO.
Although both of these ions have higher charges than the ions in lithium bromide they still balance each other in a one-to-one ratio. The chemical formula for an ionic compound represents one formula unitthe simplest ratio of the compounds positive ions cations and its negative ions anions. Li2SO4 What is the molecular formula for the compound with a formula mass of 5812 amu and an empirical formula of C2H5.
Number of atoms in each molecule b. Number of ions in each molecule c. The Correct Answer is.
The Chemical formula of a compound represents the symbols and subscripts in the lowest whole number ratio of the atoms or ions. The simplest ratio of the compounds positive and negative ions For a molecule. The chemical formula for an ionic compound represents the a.
Thus one sodium cation cancels one chloride anion resulting in the formula Na1Cl1 or NaCl. 3 Ca atoms 2 PO 4 3-ions. The formula ceCa3PO42 represents a compound with the following.
Click again to see term.

This Lab Uses The Wonderful Paperclip As A Way To Represent Elements In This Lab Students Teaching Chemistry Physical Science High School Chemistry Classroom

Molecular Compound Easy Science Molecular Ionic Bonding Molecules

Pin On Drawings For Learning Chemistry

Finding The Formula Of An Ionic Compound Worked Example Video Khan Academy
How To Decipher The Formulas Of Ionic Compounds Dummies





Post a Comment for "Chemical Formula For An Ionic Compound Represents"