Chemical Formula With Brackets Examples
Replace immutable groups in compounds to avoid ambiguity. The chemical formula for water is ceH_2O.
The chemical formula for sulfuric acid is ceH_2SO_4.

Chemical formula with brackets examples. Substitute immutable groups in chemical compounds to avoid ambiguity. Since it only appears once no brackets are needed. However use the oxidation number indicated in brackets after the cation.
100 rows 29102019 The ratio of each element is usually expressed by a chemical formula. 263 rows The chemical formula of a compound is a symbolic representation of its chemical. Brackets are only needed when the polyatomic group is greater than 1.
11022019 Water is composed of hydrogen and oxygen in a 21 ratio. Fe3 I- Fe2 I2. Hydroxide group Carbonate group Nitrate group 2.
We do this to indicate that textNO_3 is a compound ion and that there are. Also the acetic acid and formaldehyde have the same. NB 2 O 3.
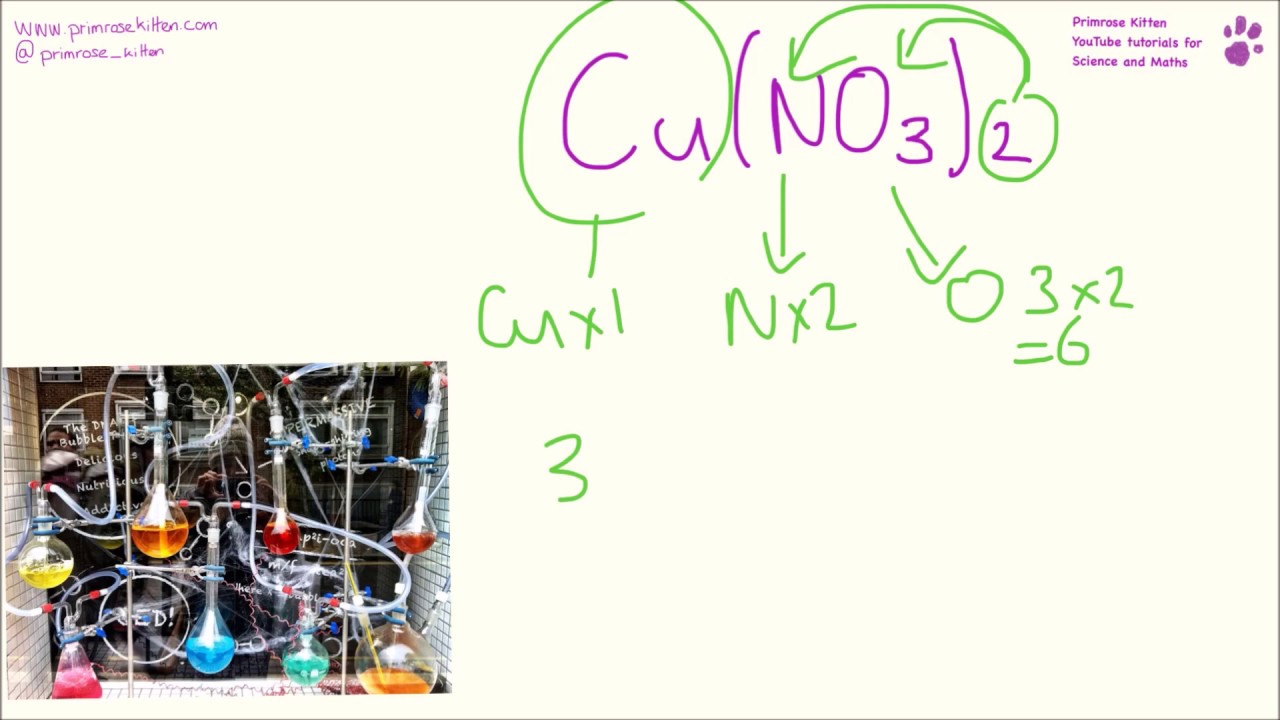
In case the number of polyatomic ion is one the bracket is not required. B Magnesium hydroxide MgOH 2. Notice how in the last example we wrote textNO_3 inside brackets.
What does this mean. H 2 Ol is liquid water but H 2 Og is steam and H 2 Os is ice. To enter an ion specify charge after the compound in curly brackets.
That is a simple HO that expresses the 11 ratio of components elements. Brackets are only used in a chemical formula if the number of polyatomic groups is greater than one. It contains the symbols of the atoms of the elements present in the compound as well as how many there are for each.
Brackets multiply whatever is inside them. The empirical formula of hexane is C 3 H 7. To enter an electron into a chemical equation use - or e.
In this example zinc will replace hydrogen from sulfuric acid to form zinc sulfate. A Sodium hydroxide NaOH. 3 or 3 or 3.
Co - cobalt and CO - carbon monoxide. Determine how many of each atom there are in the following compound. Sulfuric acid is one of the most widely produced chemicals in the United States and is composed of the elements hydrogen sulfur and oxygen.
The formula for the ammonium ion is NH4. Whenever applicable write simple ratios of the valencies. A chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule using chemical element symbols numbers and sometimes also other symbols such as parentheses dashes brackets commas and plus and minus signs.
Fe Au Co Br C O N F. But the brackets have a 2 to the right so every number inside the brackets is multiplied by two so there are two hydrogen and two oxygen atoms. Well if you see OH 2 in a formula there is no number next to the oxygen or hydrogen so there is one of each.
When are brackets needed. A chemical formula is a way of expressing information about the proportions of atoms that constitute a particular chemical compound using a single line of chemical element symbols numbers and sometimes also other symbols such as parentheses dashes and brackets. The final formula must be written without any sign of the charge.
An example for single displacement reaction is XY Z XZ Y. For example C6H5C2H5 O2 C6H5OH CO2 H2O will not be balanced but XC2H5 O2 XOH CO2 H2O will. The polyatomic group is the hydroxide OH group.
In the same way the empirical formula for hydrogen peroxide is H 2 O 2. Fe Au Co Br C O N F. Chemical name chemical formula copperI fluoride manganeseII oxide nickelII chloride tinII sulfide.
Strontium phosphate Sr3PO42 3 The combining power of silver is 1 and zinc is 2. HClg is hydrogen chloride gas but HClaq is hydrochloric acid. A chemical formula is not a chemical name and it contains no words.
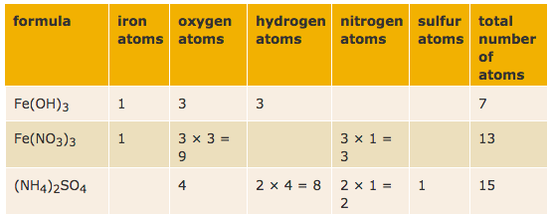
02122019 A chemical formula tells us the number of atoms of each element in a compound. Although a chemical. Except that if youre not sure how to decide whether theres only one ion in a compound or two or three then have a look here.
Follow the same rules used when writing the formula of regular binary compounds. MgOH 2 Al 2 SO 4 3 NH 4 2 SO 4 Thats it for brackets. But its molecular formula is C 6 H 14 and its structure is CH 3 CH 2 CH 2 CH 2 CH 2 CH 3.
Ionic charges are not yet supported and will be ignored. The formula for the ammonium ion is NH4 4 The combining power of silver is 1 and zinc is 2. Top tips for writing chemical formulae where youll learn about balancing the charges of positive and negative ions.
These are limited to a single typographic line of symbols which may include subscripts and superscripts. 14092018 The radical polyatomic ions must be written in brackets before the subscript is written. 02042020 In single displacement reaction any one chemical partner exchanges from reactants to products while two sets of chemical partners exchanges from reactants to products.
Chemical formula chemical name CuF 2 MnO 2 NiCl 3 SnS 2 HgI 2 ii Writing Formula 1. For example NaOH.

Counting Atoms In A Formula Chemsimplified
Brackets In Chemical Equations Youtube

Chemical Formulae What Do They Mean
Matter Atoms Chemical Formulae Brackets Pathwayz

Algebra Symbols I Math Posters Studying Math Math Methods Maths Algebra





Post a Comment for "Chemical Formula With Brackets Examples"