Chemical Equation For Copper Oxide And Carbon
Tin metal is heated with oxygen gas to yield tinIV oxide. 23092019 Chemical equation for Single displacement.

Redox Reaction In Terms Of Gain And Loss Of Oxygen Spm Chemistry
Copper oxide carbon dioxideSymbol equationCuCO3 --.

Chemical equation for copper oxide and carbon. Here copper carbonate is a reactant and copper oxide and carbon dioxide are products. Two unnamed black solids are known to be either copper II oxide or carbon. In kJ using the following thermochemical equations.
In the sieve of silver the recovery of nitrate solution involved displacement by copper metal. 2CuO C 2Cu CO2 If you do the oxidation numbers the copper in CuO has an oxidation number of 2. CaO H 2 O.
Take a look at this word equation for the reaction. Note that this is an example of a single displacement reaction. Zn s t CuSO 4 aq -.
Here is the balanced symbol equation. BaCl 2 aq K 2 SO 4 aq -. Here Ba and K displaces each other.
You should never change the formula of a substance to do this. Copper II oxide and carbon will react as follows. BaSO 4 2KCI.
Typically we would fiercely heat the copper carbonate in a vessel whose outflow was bled into a solution of limewater C aOH2 aq The liberated carbon dioxide would react with the calcium hydroxide to form a carbonate. Cu s AgNO 3aq Ag s CuNO 3 2aq In a blast furnace the reaction between solid iron III oxide and carbon monoxide gas produces solid iron and carbon dioxide gas. The following is the chemical equation showing the reactant or starting material on the left of the arrow and the products on the right of the arrow.
For each of the following generate a chemical equation. The reduction can proceed at lower temperature in a vacuum. Most carbonates undergo this decomposition provided you supply enuff heat to the reaction.
You can see that we now have two copper atoms and two oxygen atoms on each. Chemical equation for double displacement. Cu 2CO 3OH 2s 2CuO s CO 2g H 2O g The copper II oxide CuO is heated with carbon C to reduce the copper oxide to copper metal Cu as shown in the following chemical equation.
11052009 Word equationcopper II carbonate --. There are two types of chemical equations. TYPES OF CHEMICAL EQUATIONS.
Devise three or more methods of labelling them correctly using chemicals and apparatus in the laboratory test each method and compare them to decide which is best. Creative Problem Solving in Chemistry. CuOs COg --.
Cus CO2g Calculate Delta H. In this video well write the correct formula for Copper II oxide CuOTo write the formula for Copper II oxide well use the Periodic Table and follow. CO2 o 573k a 548k theoreticaL 1 k 00422 ire-i 1 o u 04 06 08 tO oc Fig.
Express your answer as a chemical equation. Copper metal can be obtained by heating copper oxide in the presence of carbon monoxide CO according to the reaction. 003 02 001 esperirnental 2.
When solid copper reacts with aqueous silver nitrate the products are copper II nitrate and solid silver. In this chemical reaction copper carbonate decomposes to form copper oxide and carbon dioxide as a products. CaOH 2 Energy.
Copper oxide is the product. 2Cu O 2 2CuO. They are balanced and un balanced Chemical reactions.
It is also known as a single displacement reaction. Ammonia gas reduces copperII oxide to copper metal and copperI oxide at 425-700 C. D dt QW 006 005 00e.
It is a decomposition reaction. ZnSO 4 aq Cus Here Zn Displaces Cu from its Solutions CuSO 4. Copper oxide carbon copper carbon dioxide 2CuO C 2Cu CO 2 Scientist Mark Miodownik shows how copper ore can be heated on an open fire to extract the metal.
01011989 Kinetics of the reduction of copper oxide with carbon monoxide 2821 d. Carbon and copper oxide. It is a decomposition reaction.
Hydrogen and carbon monoxide reduce copperII oxide to the metal at 250 C and to copperI oxide at about 150 C. Copper and oxygen are the reactants because they are on the left of the arrow. CopperII oxide is decomposed to copperI oxide and oxygen at 1030 C and atmospheric pressure.
Copper oxygen copper oxide.

Objective To Be Able To Heat A Chemical Safely Using A Bunsen Burner Ppt Download

How To Balance Cuo C Cu Co2 Youtube
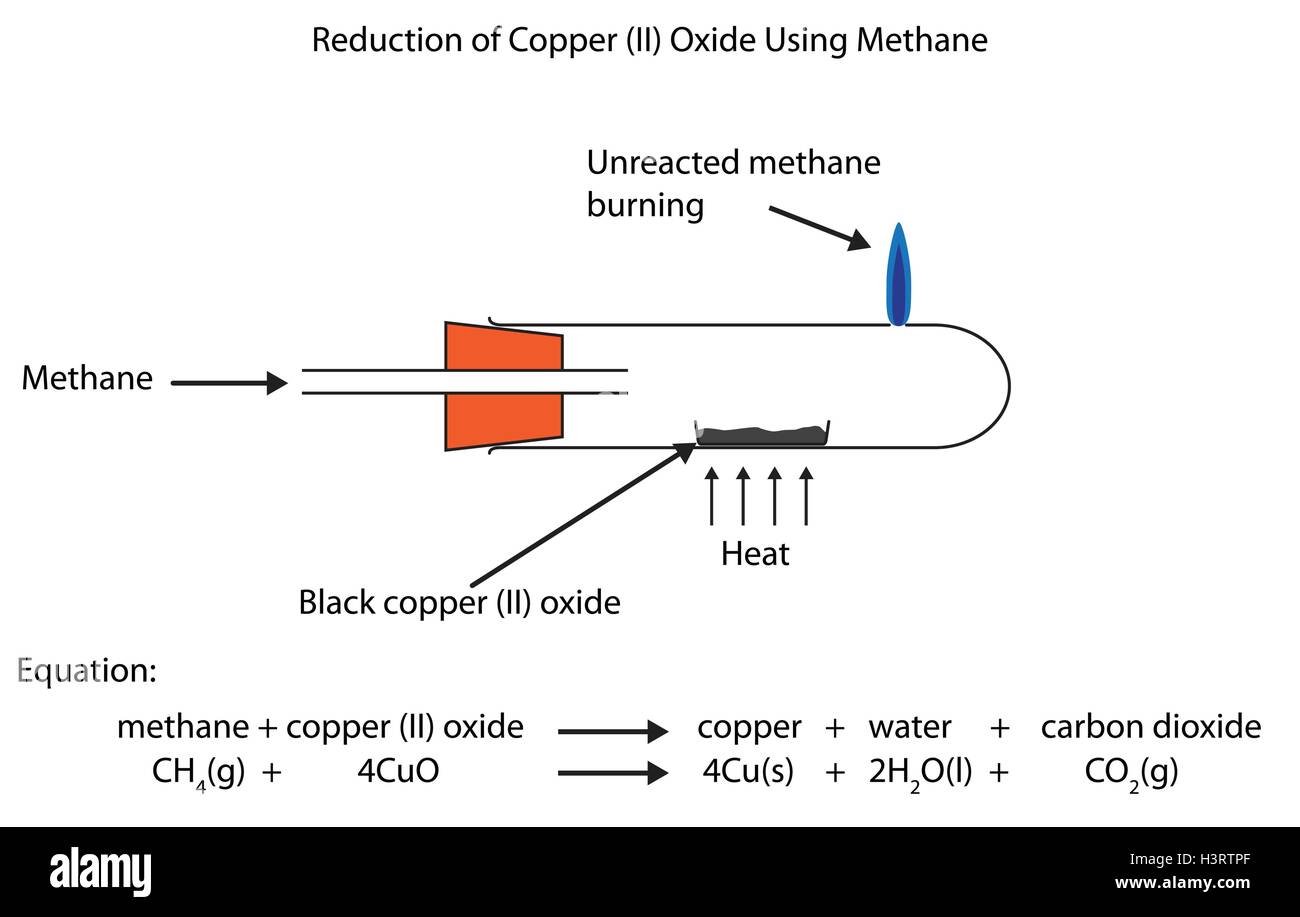
Fully Labelled Diagram Showing The Reduction Of Copper Ii Oxide Using Methane Stock Vector Image Art Alamy

When Hydrogen Is Passed Over Copper Oxide Copper And Steam Are Formed Write A Balanced Equation For This Reaction And State Which Of The Chemicals Are I Elements Ii Compounds Iii Reactants Iv Products V Metals Vi Non
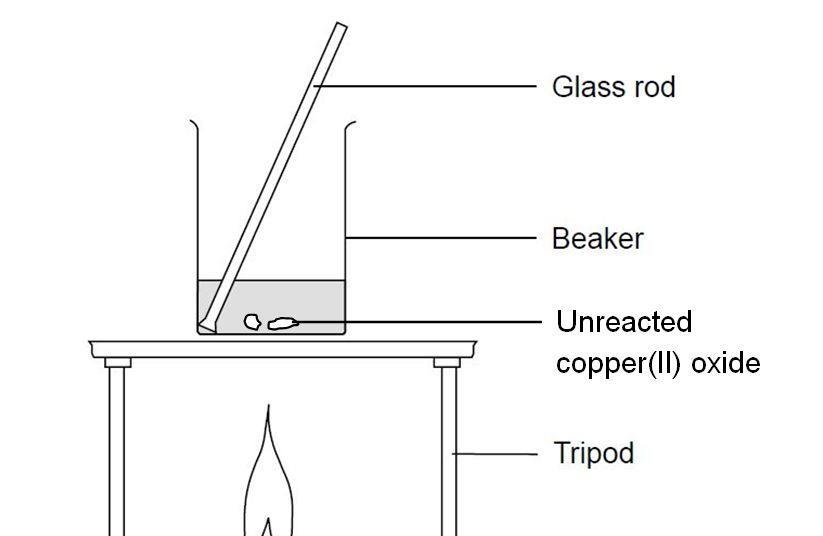
Reacting Copper Ii Oxide With Sulfuric Acid Experiment Rsc Education

Metals Learning Objectives Use Reactivity Data To Determine A Reactivity Series Relate Extraction Method To Reactivity Of Metals Write Word Symbol Equations Ppt Download
Https Chemistryklipz Files Wordpress Com 2017 01 Igcse Redox Reactivity And Analysis Pdf

Chemical Reactions Predicting Products And Balancing This Powerpoint Presentation Will Be Far More Effective If You View It As A Slideshow This Powerpoint Ppt Download
Post a Comment for "Chemical Equation For Copper Oxide And Carbon"