Chemical Equation For Calcium Nitrate And Potassium Carbonate
CaOH 2 CO 2 CaCO 3 H 2 O. 4 aM potassium Molecular H u H Total-ionic equatbn.
In The Reaction Between Aqueous Solutions Of Chegg Com
Nitric acid is an acid and potassium carbonate is a base so this is an acidbase chemical reaction.

Chemical equation for calcium nitrate and potassium carbonate. 20112012 Other examples are sodium chloride calcium chloride potassium nitrate ammonium nitrate copper sulfate magnesium sulfate calcium carbonate and sodium hydrogen carbonate. 04042018 The process is not difficult requires no specialized chemicals and the yield is excellent. Molecular Y s PCI 2 Barium chloride and sodium sulfate.
If you do not know what products are enter reagents only and click Balance. Click hereto get an answer to your question Write the balanced chemical equations for the following reactionsa Calcium hydroxide Carbon dioxide Calcium carbonate Waterb Zinc Silver nitrate Zinc nitrate Silverc Aluminium Copper chloride Aluminium chloride Copperd Barium chloride Potassium sulphate Barium sulphate Potassium chloride. The particular salt produced depends upon which acid and which alkali reacted.
Write the balanced chemical equations for the following reactions. In reality things are a. 28122017 Ca s 2H2O l Ca OH2 aq H2 g Calcium starts floating because the bubbles of hydrogen gas formed stick to the surface of the metal.
15012013 Heres the complete molecular equation for this reaction. Acid alkali salt water 4. Oversimplified description would be.
When nitric acid reacts with potassium hydroxide the salt produced is potassium nitrate. Zn 2AgNO 3 ZnNO 3 2 2Ag. Calcium hydroxide Carbon dioxide Calcium carbonate Water.
H 6 Net-ionic equation. 1 Potassium phosphate and calcium nitrate. Solid calcium carbonate reacts with hydrochloric acid HClaq to yield aqueous calcium chloride carbon dioxide gas and liquid water calcium carbonate hydrochloric acid calcium chloride carbon dioxide water CaCO 3 s 2 HClaq CaCl.
Total-ionic - 3 Sodium ana nitrate. The second step is the formation of solid calcium hydroxide as the only product from the reaction of the solid calcium oxide with liquid water. K_2CO_3_aq CaNO_3_2_aq -.
Acid alkali When an alkali and an acid react the product is a salt solution. The balanced chemical formula for this reaction is MgNO32 aq 2KOH aq MgOH2 s 2KNO3 aq. 26092017 The potassium and nitrate ions remain in a solution.
Calcium chloride nitric acid calcium nitrate hydrochloric acid CaCl2 2HNO3 CaNO32 2HCl potassium carbonate barium chloride potassium chloride barium carbonate K2CO3 BaCl2 2KCl BaCO3. In many cases a complete equation will be suggested. Chemical equation of reaction of CaNO32 K2CrO4 2KNO3 CaCrO4.
The unbalanced chemical reaction is. Potassium react violently with cold water and its reaction is so violent and exothermic that the evolved hydrogen immediately catches fire. As a result of the reaction of calcium nitrate CaNO 3 2 and potassium chromate K 2 CrO 4 produces potassium nitrate KNO 3 calcium chromate CaCrO 4.
Metal carbonate acid salt carbon dioxide water 3. Substitute immutable groups in chemical compounds to avoid ambiguity. 2K s 2H2O l 2KOH aq H2 g heat.
Potassium carbonate is K2CO3. 5 arrvmniurn Molecular equation Ba 9 9 Total-ionic Net-Onic equation. The equation in which the number of atoms of all the molecules is equal on both sides of the equation is known as a balanced chemical equation.
Calcium Nitrate Potassium Chloride Potassium nitrate Calcium Chloride. Compound states like s aq or g are not required. Write a balanced chemical equation for each step of the process.
Zinc Silver nitrate Zinc nitrate Silver. Metal salt solution. Following reaction is the precipitation reaction between aqueous solutions of calcium nitrate and potassium carbonate.
For instance equation C6H5C2H5 O2 C6H5OH CO2 H2O will not be balanced but PhC2H5 O2 PhOH CO2 H2O will. CaCO_3_s KNO_3_aq Heres the net ionic equation which leaves out spectator ions. CaNOag KCO aq2 KNO aqCaCOs ite the net ionic equation for the reaction.
A Calcium hydroxide Carbon dioxide Calcium carbonate Water b Zinc Silver nitrate Zinc nitrate Silver c Aluminium Copper chlorideAluminium chloride Copper d Barium chloride Potassium sulphate Barium sulphate Potassium chloride. The first step is the decomposition of solid calcium carbonate from seashells to form solid calcium oxide and gaseous carbon dioxide.
Solved Molecular Ionic And Net Ionic Formulas For Calci Chegg Com
1f Solutions Of Potassium Carbonate And Calcium Chegg Com
13477 34 4 Calcium Nitrate Tetrahydrate 039 00735 Detail Information Common Chemicals Lab Tools Laboratory Chemicals Fujifilm Wako Chemicals U S A Corporation

How To Balance Agno3 K2co3 Ag2co3 Kno3 Youtube

Solved Aqueous Solutions Of Lithium Sulfate And C
5 Consider The Reaction Of A Solution Of Calcium Chegg Com
Write The Balanced Chemical Equations For The Following Reactions A Calcium Hydroxide Carbon Dioxide Calcium Carbonate Water B Zinc Silver Nitrate Zinc Nitrate Silver C Aluminium Copper Chloride
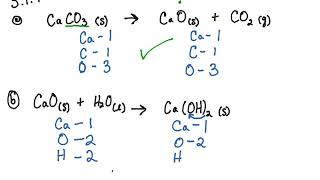
5 1 Writing And Balancing Chemical Equations Problems Chemistry Libretexts

Net Ionic Equation Writing From Names




Post a Comment for "Chemical Equation For Calcium Nitrate And Potassium Carbonate"